- Home
- Services
- Newsletter
- Products
- About
- Facility
- Management
- Team
- Scientific Advisory Board
- Corporate Info
- Quality Standards
- Employment and Diversity
- Sales Terms and Conditions
- Shipping
- Group
- Analog Devices Alliance Partner
- Client-centric philosophy
- Biosensor Research Project
- Masters Research Program - SSI
- UN Sustainable Development Goals
- NDA policy
- ZP Culture
- News and Media
- Contact/Support
- Knowledge Base
- Store
What's wrong with CBD HPLC analysis?
In the notes on this page we discuss the issue of measuring CBD and THC using chromatography.
We also link to a ZP technology for measuring CBD and THC.

Measuring THC and CBD by HPLC
The issue with HPLC analysis of CBD in products such as hemp, CBD oils etc is that there is not a single recognised international test procedure.
At the time of writing this note there is not an FDA or DEA approved method, though bodies such as the LGC in the UK are working on standardising a method. The issue is that without an internationally recognised test method every lab has a slightly different HPLC methodology and so the same sample if submitted to different labs can have dramatically different CBD and THC levels, which is not due to the cannabinoids, but rather is due to differences in the methods of analysis.
It is with this understanding that ZP offers HPLC analysis of cannabinoids, including THC and CBD.

CBD - Percentage Error 40 to - 100 %
In a recent report by the LGC in the UK a 1 % sample of CBD was sent to over 30 laboratories both regional and international; it was interesting from their report was that a number of the laboratories had gross errors when reporting the sample with range of percentage error being in the 40 to -100 percentage error range.
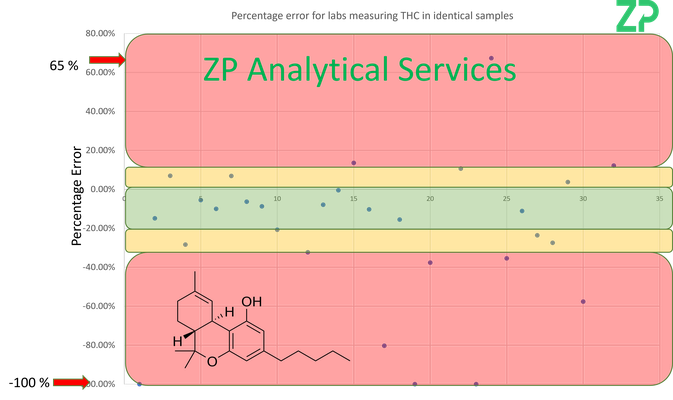
THC - Percentage Error 65 to - 100 %
The legal limit of THC in a product is approximately 0.2 % depending on jurisdiction.
The issue for growers of hemp etc is that a crop could be deemed illegal if to much THC is in the crop. Similarly products such as CBD oil have to be kept below this level.
As with CBD there is a significant lab to lab variation when different labs measure the same THC sample. In a recent LGC report, where approximately 31 labs tested the 'same' the same THC sample, the range of measured THC values had percentage errors of 65 to -100 %. The problem is that this could be enough to deem a product or crop 'illegal' , but the same sample when tested in a different lab could be reported as 'legal'.